Learn about the nursing care management of patients with dengue hemorrhagic fever in this nursing study guide.
What is Dengue Hemorrhagic Fever?
Some patients with dengue fever go on to develop dengue hemorrhagic fever, a severe and sometimes fatal form of the disease.
- Dengue fever is an acute febrile disease caused by infection with one of the serotypes of the dengue virus. It is a mosquito-borne disease caused by the genus Aedes.
- Dengue is also known as Breakbone Fever, Hemorrhagic Fever, Dandy Fever, Infectious Thrombocytopenic Purpura.
- Dengue hemorrhagic fever is a fatal manifestation of the dengue virus that manifests with bleeding diathesis and hypovolemic shock.
- These viruses are related to the viruses that cause the West Nile infection and yellow fever.
Pathophysiology
The pathophysiology of dengue hemorrhagic fever include:
- Initial phase. The initial phase of DHF is similar to that of dengue fever and other febrile viral illnesses. The virus is deposited in the skin by the vector, within few days viremia occurs, lasting until the 5th day for the symptoms to show.
- Hemorrhagic symptoms. Shortly after the fever breaks or sometimes within 24 hours before, signs of plasma leakage appear along with the development of hemorrhagic symptoms.
- Vascular leakage. Vascular leakage in these patients results in hemoconcentration and serous effusions and can lead to circulatory collapse.
- Progression. If left untreated, DHF most likely progresses to dengue shock syndrome.
Statistics and Incidences
Dengue is a reportable disease in the United States; known or suspected cases should be reported to public health authorities.
- Globally, 2.5 to 3 billion individuals live in approximately 112 countries that experience dengue transmission.
- Annually, approximately 50-100 million individuals are infected.
- Dengue fever has a mortality rate of less than 1%.
- When treated, DHF has a mortality rate of 2-5%, but when left untreated, the mortality rate is as high as 50%.
- Dengue fever may occur at any age but is more common among children.
Causes
The etiologic agent and vector of dengue:
- Flavivirus. It is caused by infection of one of the four serotypes of dengue virus, which is a Flavivirus, a genus of single-stranded nonsegmented RNA virus.
- Aedes aegypti. Dengue virus is transmitted by day-biting mosquitoes of the genus Aedes that breed in stagnant water. It has white dots at the base of its wings, with white bands on the legs.
- Incubation period. It has an incubation period of three to ten days.
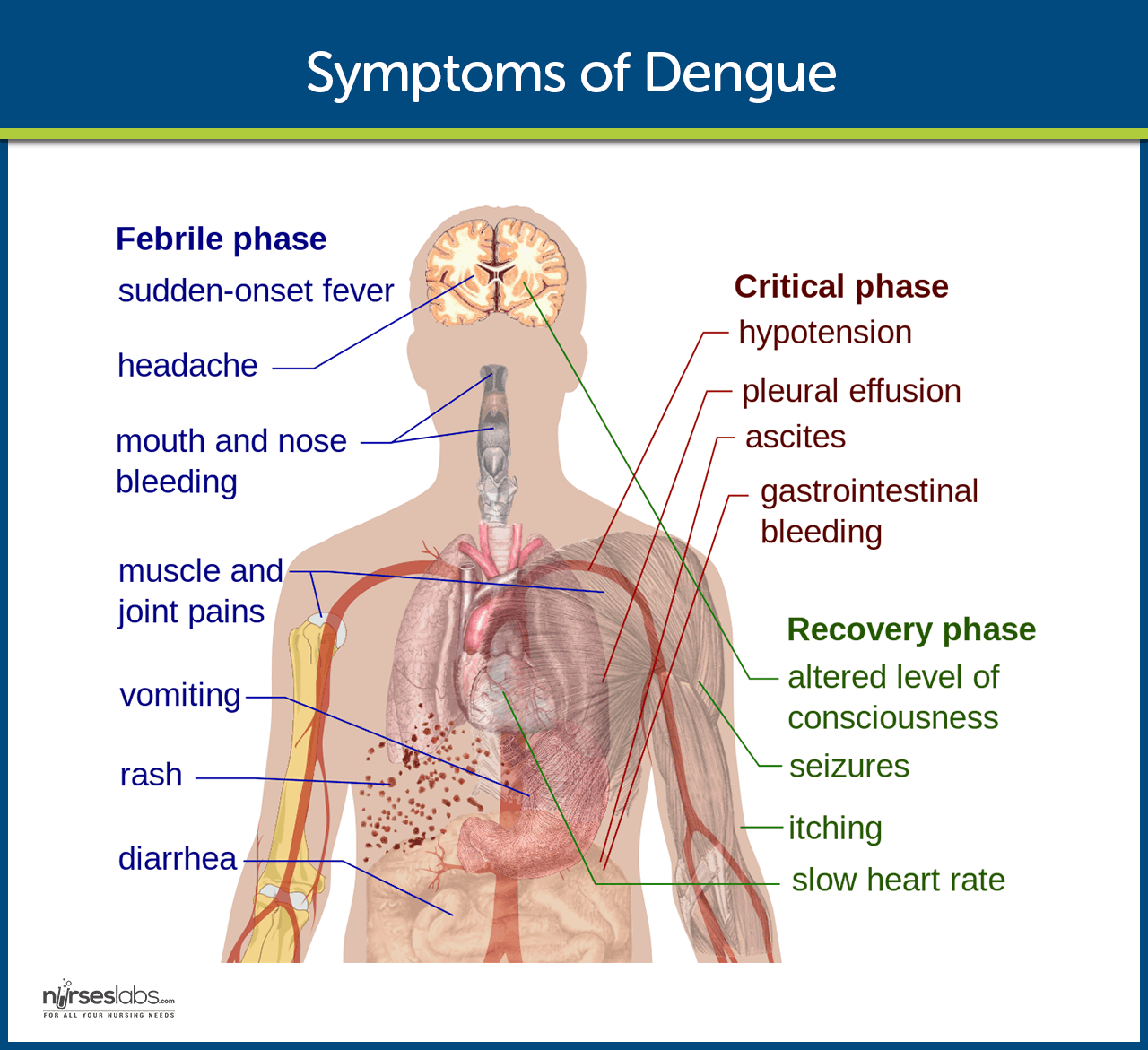
Clinical Manifestations
Symptoms, which usually begin 4 to 6 days after infection and may last to up to 10 days, include:
- High fever. Sudden high fever occurs as a result of the infection.
- Severe headaches. Severe headaches also torment the patient.
- Damage to lymph and blood vessels. As the virus slowly spreads, even the lymph and blood vessels are affected.
- Bleeding. Bleeding from the nose and gums is a characteristic of DHF.
- Enlargement of the liver. The dengue virus could also penetrate the liver, causing fatal damage.
- Circulatory system failure. The circulatory system ultimately fails eventually if the disease is not treated promptly.
Prevention
There are many ways to prevent dengue fever but there are no vaccines available yet.
- Avoid crowded places. Stay away from heavily populated residential areas.
- Mosquito repellents. Use mosquito repellents that are mild for the skin, even indoors.
- Proper clothing. When outdoors, wear long-sleeved shirts and long pants tucked into socks.
- Mosquito-free environment. Make sure window and door screens are secure and free of holes or use mosquito nets.
- Stagnant water. Empty or cover bottles, cans, and any containers with stagnant water as these can become breeding places of mosquitoes.
Complications
Dengue cases may be not adequately recognized in the United States, and as a result, many cases often end up with complications.
- Dengue shock syndrome. Common symptoms in impending shock include abdominal pain, vomiting, and restlessness.
Assessment and Diagnostic Findings
Laboratory criteria for the diagnosis of dengue virus may include 1 of the following:
- Dengue virus isolation. Isolation of the dengue virus from serum, plasma, leukocytes, or autopsy samples.
- Immunoglobulin titers. Demonstration of a fourfold or greater change in reciprocal immunoglobulin or IgM antibody titers to one or more dengue virus antigens in paired serum samples.
- Immunohistochemistry. Demonstration of the dengue virus antigen in autopsy tissue via immunohistochemistry or immunofluorescence.
- Polymerase chain reaction. Detection of viral genomic sequences in autopsy tissue, serum, or cerebrospinal fluid samples via PCR.
- Complete blood count. In DHF, there may be the presence of increases hematocrit level secondary to plasma extravasation and/or third-space fluid loss.
- Decreased platelet count. This test confirms dengue.
- Guaiac test. Guaiac testing for occult blood in the stool should be performed on all patients suspected of dengue virus infection.
Medical Management
The management of DHF is actually simple as long as it is detected early.
- Oral rehydration therapy. Oral rehydration therapy is recommended for patients with moderate dehydration caused by high fever and vomiting.
- IV fluids. IVF administration is indicated for patients with dehydration.
- Blood transfusion and blood products. Patients with internal or gastrointestinal bleeding may require transfusion, and patients with coagulopathy may require fresh frozen plasma.
- Oral fluids. Increase in oral fluids is also helpful.
- Avoid aspirins. Aspirin can thin the blood. Warn patients to avoid aspirins and other NSAIDs as they increase the risk for hemorrhage.
Nursing Management
Nursing management of patients with DHF is essential in achieving complete recovery.
Nursing Assessment
Assessment of a patient with DHF should include:
- Evaluation of the patient’s heart rate, temperature, and blood pressure.
- Evaluation of capillary refill, skin color and pulse pressure.
- Assessment of evidence of bleeding in the skin and other sites.
- Assessment of increased capillary permeability.
- Measurement and assessment of the urine output.
Nursing Diagnosis
Based on the assessment data, the major nursing diagnoses for a patient with DHF are:
- Risk for bleeding related to possible impaired liver function.
- Deficient fluid volume related to vascular leakage.
- Pain related to abdominal pain and severe headaches.
- Risk for ineffective tissue perfusion related to failure of the circulatory system.
- Risk for shock related to dysfunction in the circulatory system.
Nursing Care Planning and Goals
The goals in a patient with DHF are:
- Be free of signs of bleeding.
- Display laboratory results within normal range for individuals.
- Maintain fluid volume at a functional level.
- Report pain is relieved or controlled.
- Follow prescribed pharmacologic regimen.
- Demonstrate adequate tissue perfusion.
- Display hemodynamic stability.
- Be afebrile and free from other signs of infection.
Nursing Interventions
Nursing interventions appropriate for a patient with DHF include:
- Blood pressure monitoring. Measure blood pressure as indicated.
- Monitoring pain. Note client report of pain in specific areas, whether pain is increasing, diffused, or localized.
- Vascular access. Maintain patency of vascular access for fluid administration or blood replacement as indicated.
- Medication regimen. There must be a periodic review of the medication regimen of the client to identify medications that might exacerbate bleeding problems.
- Fluid replacement. Establish 24-hour fluid replacement needs.
- Managing nose bleeds. Elevate position of the patient and apply ice bag to the bridge of the nose and to the forehead.
- Trendelenburg position. Place the patient in Trendelenburg position to restore blood volume to the head.
Evaluation
A successful nursing care plan has achieved the following:
- Absence of signs of bleeding.
- Displayed laboratory results within normal range for individuals.
- Maintained fluid volume at a functional level.
- Reported pain is relieved or controlled.
- Followed prescribed pharmacologic regimen.
- Demonstrated adequate tissue perfusion.
- Displayed hemodynamic stability.
- Afebrile and free from other signs of infection.
Discharge and Home Care Guidelines
A patient with DHF discharged from the health care facility should be instructed to:
- Avoid diuretics. Avoid caffeine and alcohol as indicated to reduce effects of diuresis.
- Follow-up appointments. Comply with the recommended medical and laboratory follow-ups.
- Oral care. Recommend use of soft toothbrush to reduce risk of injury to the oral mucosa.
- Diet. Foods rich in vitamin K should be recommended to promote blood clotting.
- Education. Educate patient on the use of mosquito nets and insecticides.
Documentation Guidelines
The focus of documentation in a patient with DHF include:
- Factors that potentiate blood loss.
- Baseline vital signs, mentation, urine output, and subsequent assessments.
- Results of laboratory and diagnostic studies.
- Degree of deficit and current sources of fluid intake.
- I&O and fluid balance.
- Client’s description of response to pain and acceptable level of pain.
- Plan of care.
- Teaching plan.
- Response to interventions, teachings, and actions performed.
- Attainment or progress towards desired outcomes.
- Modifications to plan of care.





























Leave a Comment